
Olefin Metathesis
Download a copy of Olefin Metathesis here
Author: Dr Joseph Cowell
Olefin metathesis is the reaction between two olefin moieties in the presence of a metal catalyst to form two new olefin species. Although the reaction itself has been known and used since the 1950s, the exact mechanism for this reaction wasn’t well understood until much more recently.

It wasn’t until the pioneering work of Yves Chauvin, Robert Grubbs, and Richard Schrock that the synthetic utility of this reaction could be fully realised. In 1971 Yves Chauvin proposed a catalytic cycle with a metal carbene as the catalyst which proceeded through a metallocyclobutane intermediate. Chauvin’s mechanism suggested metal-alkylidene complexes should be the ideal catalysts for olefin metathesis which allowed for a more focused search for metathesis catalysts.

This led Richard Schrock to synthesise the first commercially available molybdenum catalyst for metathesis in 1990.1 Although this molybdenum catalyst is still the most reactive metathesis catalyst now, it is not without its limitations. High sensitivity to water and oxygen, along with a lack of functional group tolerance, limits its use in the lab. As such, Robert Grubbs developed a series of more stable ruthenium-based catalysts in 1995 that showed much better water and oxygen stability and significantly better functional group tolerance.2 Although many further metathesis catalysts have since been developed, it was these initial molybdenum and ruthenium catalysts from Schrock and Grubbs that paved the way for olefin metathesis being the synthetic tool it is today.
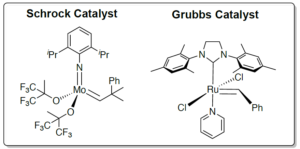
For their work, the Nobel Prize in Chemistry in 2005 was awarded jointly to Yves Chauvin, Robert H. Grubbs, and Richard R. Schrock “for the development of the metathesis method in organic synthesis.”3

The development of easy-to-handle olefin metathesis catalysts led to a wide range of metathesis reactions being incorporated into both important large scale industrial processes, and the synthesis of small molecule pharmaceuticals.
One of the largest industrial processes that olefin metathesis is used in, is the Shell Higher Olefin Process (SHOP). This process synthesises linear olefins for use as detergent alcohols and detergent alkylates.4 The SHOP process proceeds via 2 steps, firstly an oligomerisation step, and then an olefin metathesis step which uses an alumina supported molybdenum catalyst. As an example of its importance, this process produces over 1 million tonnes of olefins per year.
Metathesis has also found a use in the pharmaceutical industry where it is now routinely used in the synthesis of new pharmaceuticals. One such example is Simeprevir which was approved for the treatment of Hepatitis C by the European Union in 2014. One of the key steps in the synthesis of Simeprevir is a ring closing olefin metathesis reaction which uses 1st generation Hovyeda-Grubbs catalyst to form a new 14-membered ring.6

References
- R. Schrock, J.S. Murdzek, G.C. Barzan, J. Robbins, M. DiMare and M. O`Regan, J. Am. Chem. Soc., 1990, 112, 3875.
- Schwab, M.B. France and J.W. Ziller, R.H. Grubbs, Angew. Chem., Int. Ed. Engl., 1995, 34, 2039.
- https://www.nobelprize.org/uploads/2018/06/advanced-chemistryprize2005.pdf
- C. Mol, Journal of Molecular Catalysis A: Chemical, 2004, 213, 39.
- A. McAuley et al., J. Med. Chem., 2010, 53, 2443.
- Rosenquist et al., J. Med. Chem., 2014, 57, 1673
